Barium Hydroxide
Barium hydroxide is a hydroxide of Barium. It is also known as Barium dihydroxide or Caustic Baryta.
Barium hydroxide chemical formula Ba(OH)₂. It is available in various forms such as monohydrate, and octahydrate. It is used in multiple applications, especially in analytical and organic chemistry.
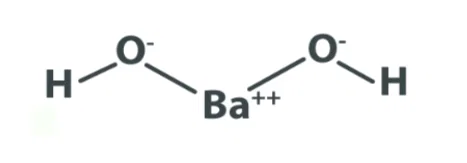
Structure– Barium hydroxide structure is formed with two hydroxyl anions (OH-) and one barium cation Ba²⁺.
Synthesis – Barium hydroxide is produced by reacting Barium chloride with sodium hydroxide in an aqueous solution.
2 NaOH (aq) + BaCl₂ (aq) → Ba(OH)₂ (s) + 2 NaCl (aq) Barium dihydroxide can also be produced by reacting Barium nitrate with Sodium hydroxide.
2 NaOH + Ba(NO₃)₂ → 2 NaNO₃ + Ba(OH)₂

Properties
Barium hydroxide monohydrate
- Molecular Formula: Ba(OH)₂·H₂O
- CAS Number: 22326-55-2
- Molecular Weight: 189.3925 gm/mol
- Appearance: White Solid
- Odour: Odourless
- Melting Point: 300°C
- Density: 3.743 gm/cm³
Barium hydroxide octahydrate
- Molecular Formula: Ba(OH)₂·8H₂O
- CAS Number: 12230-71-6
- Molecular Weight: 315.4636 gm/mol
- Appearance: Transparent crystals or white masses
- Odour: Odourless
- Melting Point: 78°C
- Density: 2.18 gm/cm³
Uses
Barium hydroxide
- It is used in the titration of weak acids (especially organic acids) in analytical chemistry.
- It is used as a strong base in organic synthesis (Example: Hydrolysis of esters and nitriles).
- It is used as an additive in rayon, thermoplastics (such as phenolic resins), and polyvinyl chloride stabilizers to improve plastic properties.
- It is used in fat saponification, fusing of silicates, sugar fabrication, and soap manufacturing.
- It is used for dehydration and deacidification, especially for removing sulphuric acid from fats, glycerol, oils, and waxes.
- It is also used in the chemical synthesis of other barium compounds and organic compounds.
Barium hydroxide monohydrate
- In water purification, it is used to neutralize acidic substances, facilitating the removal of impurities and contaminants from water sources.
- It is used as a polyvinyl chloride stabilizer for the improvement of plastics.
- It is used as a multifunctional additive in the petroleum industry.
- It can be used for barium-based lubricating grease and refining oils.
- It is used for producing barium salts and organic compounds.
- It is also used in artificial silk, glass, and enamel industries.
Barium hydroxide octahydrate
- It is used in the manufacture of alkali, glass, in synthetic rubber vulcanization, alkalizing agent in water softening, boiler scale remedy, and softening water.
- It is used in the centromeric heterochromatin banding technique.
- It acts as a precursor to prepare barium phosphate, barium sulfate, barium sulfide, and barium titanate.
- It is used in analytical chemistry for the titration of weak acids, especially organic acids.
- It is involved in the preparation of cyclopentanone, diacetone alcohol, and D-Gulonic gamma-lactone.
- It is used in ester hydrolysis and decarboxylation of amino acids.
Vishnupriya Chemicals Private Limited is one of India’s best manufacturers, suppliers, and exporters of Barium Hydroxide.
Click here for Barium Dihydroxide Grades.
Contact us for more information on Barium Hydroxide prices.

